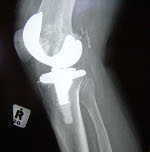
 The implant is designed to be a more cost-effective alternative to traditional orthopedic joint replacement technologies, according to the report.
The implant is designed to be a more cost-effective alternative to traditional orthopedic joint replacement technologies, according to the report.
NovoSource was founded in 2012 with monetary support from the Dayton Development Coalition, which is led by Harold Linville, former chairman for Innovative Medical Device Solutions and current NovoSource chairman and CEO. The company is also developing the NovoHip implant to market this year.
More Articles on Devices:
Medtronic Spine Launches Device for Multilevel Spinal Fusion
Wenzel Spine Launches Cervical Interbody Fusion Implant
Spine Wave Intervertebral Body Fusion Device Receives FDA 510(k) Clearance
At the Becker's 23rd Annual Spine, Orthopedic and Pain Management-Driven ASC + The Future of Spine Conference, taking place June 18–20 in Chicago, spine surgeons, orthopedic leaders and ASC executives will come together to explore minimally invasive techniques, ASC growth strategies and innovations shaping the future of outpatient spine care. Apply for complimentary registration now.
